Lab of molecular – structural biology
The lab of molecular structural biology, headed by Associate Professor Tali Haran investigates the recognition mechanisms of DNA by regulatory proteins. Our research probes the mechanism of Protein-DNA interaction in cellular function. For the last years the research focus on p53 – an important protein known for its tumor suppressor activities. Thus the research outcome may benefit diagnostic and therapy for cancer.
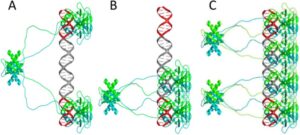
Models of p53CT tetramers (A and B) and a p53 octamer (C) bound to DNA.
Central research topic:
The identity of eukaryotic cells is defined by the correct temporal and spatial expression of specific genes. The first step in the selective expression of any gene is the ability to single it out from among all the others genes in the genome. This ability, which lies at the heart of many cellular processes, invariably requires the interactions of proteins with DNA molecules.
How do proteins recognize their specific binding sites?
Protein–DNA interactions proceed through an induced-fit mechanism, similar to the induced-fit mechanism of enzyme action. Therefore, both the DNA and the protein are not passive players, but have active roles, dictated by their structural plasticity. We use cutting edge high-throughput techniques (Selex-seq, combined with next-generation sequencing) to study protein/DNA interactions together with novel biophysical techniques to study the mechanical properties of the DNA double helix.
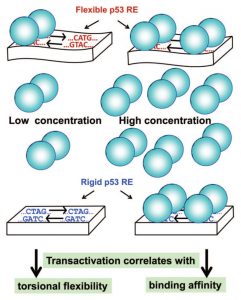
Model of p53 bound to DNA at low (left) vs. high (right) p53 concentrations.
p53 dimers are schematically represented by two connected balloons. The DNA response element (RE) is represented by a rectangular block when the DNA is rigid (containing the rigid CTAG motif) or by a wavy-rectangular block when the DNA is flexible (containing the flexible CATG motif).